


Countries
we are present in
Years
of Experience
Established in the year 2007, with a thought of “Supporting Life Worldwide”. With headquarter at Ahmedabad, currently we are in 100% export business. We have committed ourselves to provide affordable and innovative healthcare globally. Read More...
We at BioMatrix always emphasize on Trust, Quality, Integrity and Excellence in all our practices.
The company has demonstrated a robust revenue growth over the past five years .
BioMatrix’s manufacturing facility is equipped with the latest and the best of equipment, technology and manpower. Having the coveted WHO-GMP,EU-GMP and Health Canada approval, the unit is also compliant with US-FDA, UK-MHRA and ANVISA standards. Located at Ahmedabad, India, the plant has captive and buffer capacities which can meet the future demands, keeping in mind the company’s ambitious growth and expansion plans. Likewise, the design and installed machinery are also futuristic and are on par with any global manufacturing setup.
The scope and scale of our manufacturing unit help us to provide safe, affordable and effective medicines globally.

World Class R&D Facility
"Wherever the art of medicine is loved, there is also a love for humanity"
–
Hippocrates
Realising the urgency to ramp up innovation capabilities, BioMatrix has established its state-of-the-art R&D centre equipped with the latest research infrastructure and manned by domain-leading personnel. Frontline scientists, researchers, technicians, data analysis professionals, medical practitioners... They form the core BRC team which is continuously engaged in pursuit of "lab-to-life" research innovations.
BioMatrix has a strong and unwavering commitment to work ethics and business transparency. The company is well-respected and praised for its values and code of conduct. These are vital factors to consider while scouting for an international business partner.
WHO-GMP
Manufacturing Facility
European Union GMP
Certified

Health Canada
Certified
Countries
we are present in
Years of
Experience
Product
Registrations
Robust R&D
Infrastructure
Expert Regulatory
Team
Three-Star
Export House
We have miles to go and milestones to surpass.
BioMatrix's journey so far is as eventful as it is exciting. Overcoming untold challenges, we
have come thus far, and the journey continues!
What is more important, the journey or the destination?
To us, both matter equally, as we
move ahead. With zeal and determination, we aim to achieve these goals in the coming years:
UK-MHRA & TGA approval for our Manufacturing facility and commence operations in Europe, Central, South and Latin America. Expansion of our QC & Production Capabilities.
Operation of API Manufacturing Unit.
To commence a state-of-the-art manufacturing unit for injectables.
USFDA approval for our manufacturing facility.

Apixaban Tablets
2.5 mg/5 mg

Sitagliptin Tablets
25 mg/50
mg/100 mg

Metformin & Sitagliptin Tablets
500
mg/50 mg , 1000 mg/50 mg

Rivaroxaban Tablets
10 mg/15 mg/20
mg

Ticagrelor Tablets
60 mg/ 90 mg

Empagliflozin Tablets
10 mg/25 mg

Empagliflozin & Metformin Tablets
5/500 mg, 5/850 mg, 5/1000 mg

Empagliflozin & Metformin Tablets
12.5/500 mg, 12.5/850 mg, 12.5/1000 mg

Dapagliflozin Tablets
5 mg / 10 mg

Dapagliflozin & Metformin Tablets
5/500 mg, 5/850 mg, 5/1000 mg
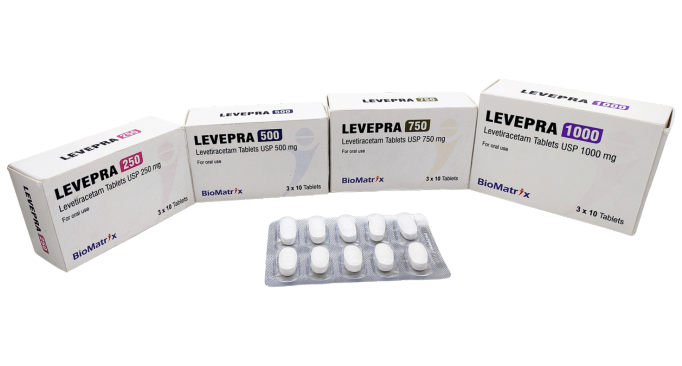
Levetiracetam Tablets
250 mg / 500 mg / 750 mg / 1000 mg
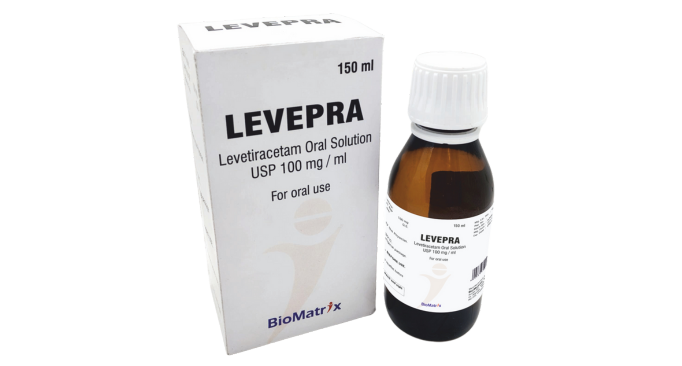
Levetiracetam Oral Solution
100 mg/ml (150 ml,300 ml)

Telmisartan Tablets
20 mg / 40 mg / 80 mg
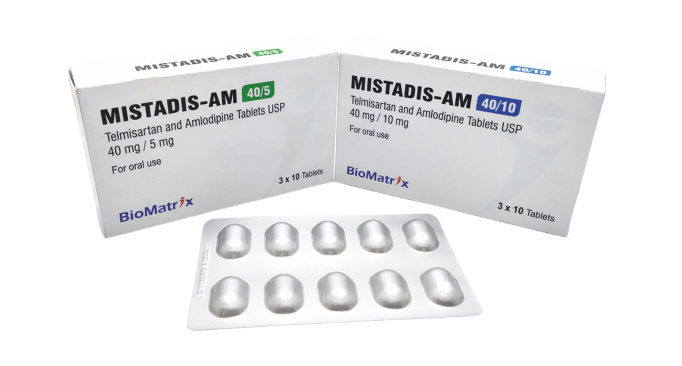
Telmisartan & Amlodipine Tablets
40/5 mg, 40/10 mg

Telmisartan & Amlodipine Tablets
80/5 mg, 80/10 mg
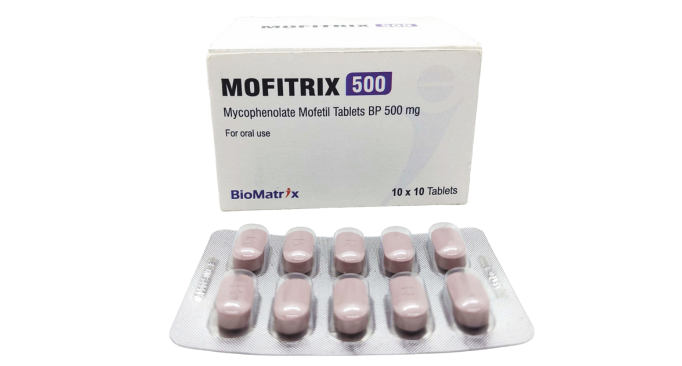
Mycophenolate Mofetil Tablets
500 mg

We are thrilled to announce that our Oral Solids, Liquids and Dry syrup facility has received EUGMP approval! This achievement is a testament to unwavering dedication and commitment of our Team.
Read more
It was a phenomenal experience for BioMatrix to be a part of Iphex 2024 and to interact with multiple international delegates to explore collaboration opportunities for improved healthcare. IPHEX, organised by PHARMEXCIL is one of the largest exhibitions showcasing Indian pharmaceutical products and technologies to a global audience.
Read more
We had a great time attending and exhibiting at CPHI Worldwide in Milan. It’s been an amazing three days of networking, learning and growth. A huge thank you to everyone who visited us and to our dedicated team for their tireless efforts that made all of this possible. We’re excited about the future collaborations and innovations ahead. Stay connected with us for more updates and exciting projects.
Read more
Exciting Milestone Achieved! We’re thrilled to share that BioMatrix Healthcare Pvt. Ltd. has received site approval from Health Canada! A big kudos to our incredible team for their unwavering dedication and expertise. This milestone reflects our commitment to quality and innovation in healthcare! Here’s to many more milestones ahead!.
Read more
Exciting Milestone Achieved! We're thrilled to share that BioMatrix Healthcare Pvt. Ltd. has received approval from KMCA – Kurdistan Medical Control Agency! A big kudos to our incredible team for their unwavering dedication and expertise. This milestone strengthens our presence in the Kurdistan region and reflects our continued commitment to quality and regulatory excellence. Here's to many more milestones ahead.
Read more
BioMatrix had an incredible experience at Iphex 2025, engaging with international delegates and exploring impactful collaborations for a healthier future. This year’s participation gave us the opportunity to strengthen global connections, showcase our commitment to quality healthcare, and open new doors for partnerships across continents.
Read more
We proudly announce a regulatory milestone with site approval from the Food and Drug Administration (FDA), Philippines.This approval reflects our continued commitment to maintaining global quality standards and ensuring the safety and efficacy of our products.
Read more
We are pleased to announce the successful approval from the Iraqi Ministry of Health, marking another important step in our global regulatory expansion. This achievement strengthens our presence in the Middle East and reflects our commitment to meeting international quality and regulatory standards.
Read more
BioMatrix Healthcare marked a strong presence at CPHI Frankfurt 2025, engaging with global leaders and collaborators to drive innovation and excellence in pharmaceuticals.
Read more
We are proud to announce the approval of our first European Market Authorization (MA) for Sitagliptin Tablets (25 mg, 50 mg & 100 mg). This achievement marks a major step forward in our global expansion journey and reflects the hard work, dedication, and excellence of our entire team.
Read more
Exciting Milestone Achieved! We're thrilled to share that BioMatrix Healthcare Pvt. Ltd. has received approval from BoMRA – Botswana Medicines Regulatory Authority! A big kudos to our incredible team for their unwavering dedication and expertise. This milestone marks another step forward in expanding our global footprint and delivering high-quality healthcare solutions worldwide. Here's to many more milestones ahead!
Read more
Exciting Milestone Achieved! BioMatrix Healthcare Pvt. Ltd. is proud to announce that Empagliflozin Tablets (10 mg & 25 mg) have received EU approval via the Decentralized Procedure (DCP). This significant achievement enables BioMatrix to obtain simultaneous marketing authorizations across multiple European Union member states, further strengthening our presence in regulated global markets.
Read more
We’re proud to announce that BioMatrix Healthcare Pvt. Ltd. has received site approval from Emirates Drug Establishment (EDE), UAE. This significant achievement marks another important step in strengthening our presence in regulated international markets and reflects our unwavering commitment to quality, compliance, and excellence in healthcare manufacturing.
Read more